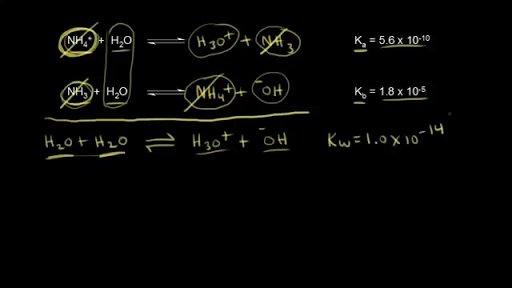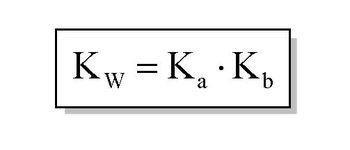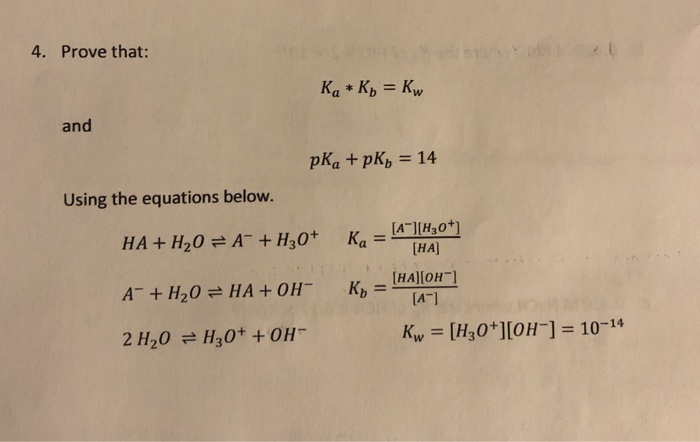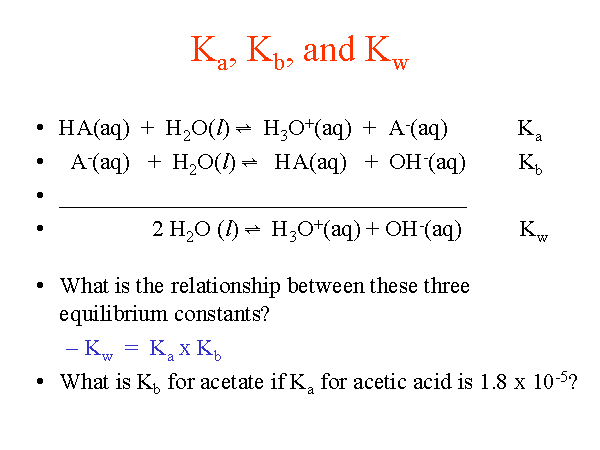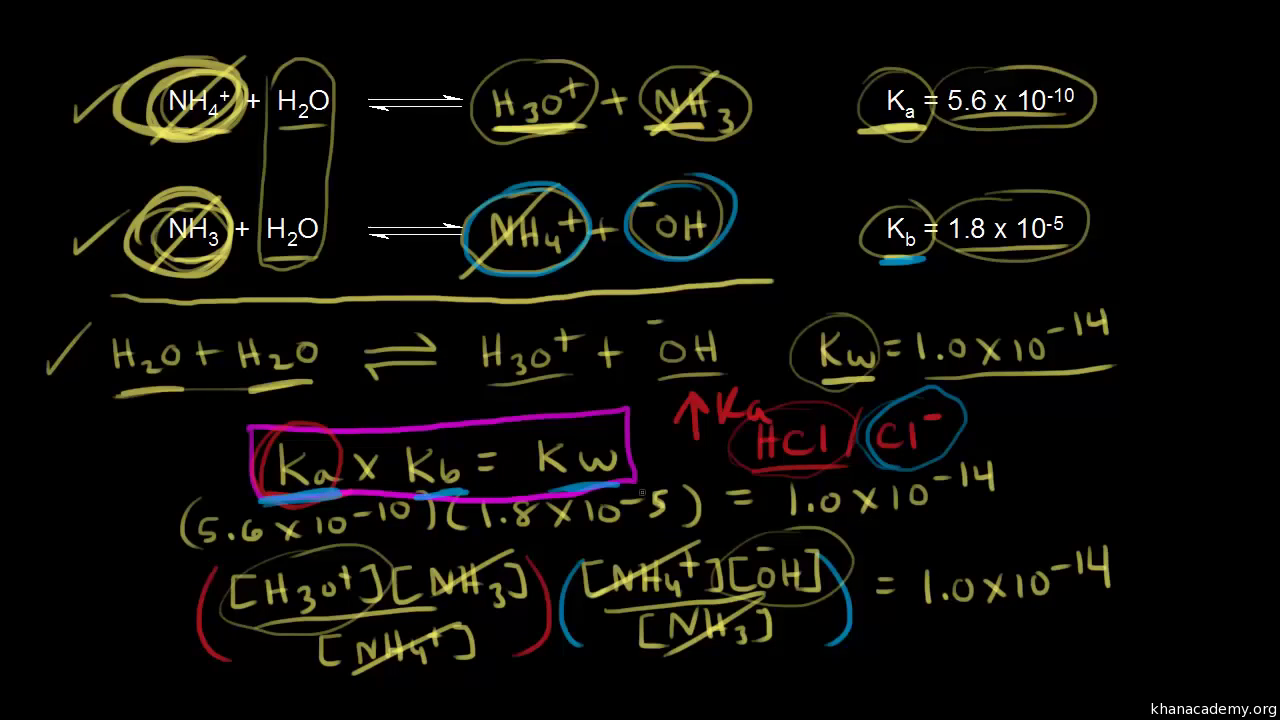NEET UG - Ionic equilibrium 09 Relation between ka, kb and kw and based numerical Offered by Unacademy

How is Kw=Ka x Kb if each of those equilibrium constants include the concentration of another substance in them? Kw=H3o x oh but ka which is h3o also includes another substance? :